An Understanding of EUnetHTA HTA

The EUnetHTA Executive Board recently adopted the following Understanding of EUnetHTA HTA. This text is the result of several months of consultation with the project’s board. The EUnetHTA Executive Board agreed that HTA in the context of EUnetHTA activities is understood to be composed of the following elements: Assessments should inform decision-making. Assessments are not decision-making processes themselves. Information should […]
OTJA10 – Final Assessment report and related documents are now available
EUnetHTA started an “Error reporting and correction” procedure on 04/05/2020. The reported error was not classified as an error by EUnetHTA and the Assessment Team, therefore no changes to the assessment report were needed. The comments received including the answers from the authoring team are published for transparency reasons. See here. We are pleased to […]
The EUnetHTA Prioritisation List (EPL) 2.0 is now published and available for access

EUnetHTA is pleased to announce the publication of the second iteration of the EPL. As part of the overarching Topic Identitication, Selection and Prioritisation (TISP) task group, the EPL is a valuable strategic tool for companies to identify the significant interest of national HTA bodies in relation to specific listed compounds. EUnetHTA seeks to increase […]
The public consultation of the draft methodological guideline “Practical considerations when critically assessing economic evaluations” is OPEN (deadline Oct. 11, 2019)

We are pleased to announce that, as of today, the draft methodological guideline “Practical considerations when critically assessing economic evaluations”, produced within WP6 B2, has entered the public consultation phase. This consultation will be open until 11th October, 2019. Objective of the methodological guideline EUnetHTA´s methodological guidelines are targeted to the needs of health […]
Public consultation on the guideline ”Process of information retrieval for systematic reviews and health technology assessments on clinical effectiveness” in August/September 2019

The methodological guideline ”Process of information retrieval for systematic reviews and health technology assessments on clinical effectiveness” is currently under revision by IQWiG, NIPHNO and AETSA. It will be made available for public consultation as of beginning of August for the duration of approximately 2 months.
Reminder: Open Call for Patient Input – Joint Assessment on a medicinal product for neovascular (wet) age-related macular degeneration (AMD).
This is a friendly reminder that EUnetHTA recently started a new Joint Assessment on a medicinal product for neovascular (wet) age-related macular degeneration (AMD). To find out about participation, please read more here. Please be advised that input submissions will be received through 13:00, July 15th.
OTCA15 – Final Assessment and input from external experts and manufacturers are now available
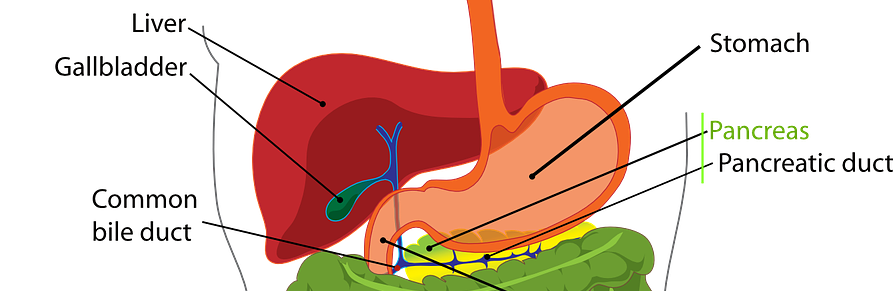
We are pleased to announce that the final assessment for OTCA15 “Irreversible electroporation in liver and pancreatic cancer”, together with the input from external experts and manufacturers, are now available for access. Irreversible electroporation (IRE) is a nonthermal ablative method based on the application of short high-voltage and low-frequency electric fields to destroy tissue. The purpose of […]
EUnetHTA Magazine – Summer 2019 – Now available

We are pleased to roll out the next issue of EUnetHTA Magazine. As we are now experimenting with a web-based format, we hope to provide a more interactive experience for readers, whether that’s on desktop, tablet, or mobile device. In this issue we have healthcare thoughts from Finland, updates from MD and an interview with […]
”Patient Input in Relative Effectiveness Assessments” is now available.

EUnetHTA is pleased to announced that the document on “Patient Input in Relative Effectiveness Assessments” is now available. The document describes the process regarding direct patient input in REAs within JA3. The document is primarily intended for those who design and conduct EUnetHTA REAs, although it may be informative for a wider audience of patients, […]
HTAi 2019 Annual Meeting – Cologne!

This year’s HTAi Annual Meeting will take place from 15 to 19 June in Cologne, Germany. The meeting brings together HTA experts from across the world for five days of discussions, panels, presentations and more. This year’s theme “HTA beyond 2020: Ready for the New Decade” focusses on what the future development of HTA worldwide looks like as a number of prominent strategies and […]

