Activities

An Understanding of EUnetHTA HTA
The EUnetHTA Executive Board recently adopted the following Understanding of EUnetHTA HTA. This text is the result of several months of consultation with the project’s
OTJA10 – Final Assessment report and related documents are now available
EUnetHTA started an “Error reporting and correction” procedure on 04/05/2020. The reported error was not classified as an error by EUnetHTA and the Assessment Team,

The EUnetHTA Prioritisation List (EPL) 2.0 is now published and available for access
EUnetHTA is pleased to announce the publication of the second iteration of the EPL. As part of the overarching Topic Identitication, Selection and Prioritisation (TISP)

The public consultation of the draft methodological guideline “Practical considerations when critically assessing economic evaluations” is OPEN (deadline Oct. 11, 2019)
We are pleased to announce that, as of today, the draft methodological guideline “Practical considerations when critically assessing economic evaluations”, produced within WP6 B2, has

Public consultation on the guideline ”Process of information retrieval for systematic reviews and health technology assessments on clinical effectiveness” in August/September 2019
The methodological guideline ”Process of information retrieval for systematic reviews and health technology assessments on clinical effectiveness” is currently under revision by IQWiG, NIPHNO and
Reminder: Open Call for Patient Input – Joint Assessment on a medicinal product for neovascular (wet) age-related macular degeneration (AMD).
This is a friendly reminder that EUnetHTA recently started a new Joint Assessment on a medicinal product for neovascular (wet) age-related macular degeneration (AMD). To
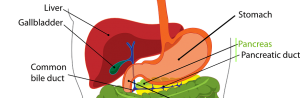
OTCA15 – Final Assessment and input from external experts and manufacturers are now available
We are pleased to announce that the final assessment for OTCA15 “Irreversible electroporation in liver and pancreatic cancer”, together with the input from external experts

EUnetHTA Magazine – Summer 2019 – Now available
We are pleased to roll out the next issue of EUnetHTA Magazine. As we are now experimenting with a web-based format, we hope to provide

”Patient Input in Relative Effectiveness Assessments” is now available.
EUnetHTA is pleased to announced that the document on “Patient Input in Relative Effectiveness Assessments” is now available. The document describes the process regarding direct

HTAi 2019 Annual Meeting – Cologne!
This year’s HTAi Annual Meeting will take place from 15 to 19 June in Cologne, Germany. The meeting brings together HTA experts from across the world for five days

